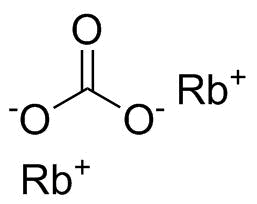
Rubidium carbonate is an inorganic compound with the chemical formula Rb2CO3. It is a white, hygroscopic crystalline salt composed of rubidium cations (Rb+) and carbonate anions (CO32−). As an alkali metal carbonate, it exhibits strong basic character in aqueous solution and behaves similarly to other group 1 carbonates such as sodium carbonate and potassium carbonate, though it is far less commonly encountered due to the relative scarcity of rubidium.
The compound consists of two rubidium ions associated with a planar carbonate ion. The carbonate ion itself has a trigonal planar geometry, with delocalized bonding over the three oxygen atoms, which contributes to its stability. In the solid state, rubidium carbonate forms an ionic lattice stabilized by electrostatic interactions between the large Rb+ cations and the doubly charged carbonate anions.
Rubidium carbonate is typically produced by reacting rubidium hydroxide or rubidium oxide with carbon dioxide. The reaction of rubidium hydroxide with carbon dioxide proceeds readily in aqueous solution or even upon exposure to atmospheric CO2, forming the carbonate salt and water. This behavior is characteristic of strong alkali metal bases, which readily absorb carbon dioxide from air to form carbonate species.
Historically, rubidium compounds were first studied following the element’s discovery in 1861 by Robert Bunsen and Gustav Kirchhoff using flame spectroscopy. While early investigations focused on identifying its spectral properties, subsequent work explored its chemistry in relation to other alkali metals. Rubidium carbonate, like other rubidium salts, has been of interest primarily in research settings rather than large-scale industrial applications due to limited availability and higher cost.
In aqueous solution, rubidium carbonate dissociates completely into rubidium ions and carbonate ions. The carbonate ion undergoes hydrolysis, producing hydroxide ions and thus creating a basic solution. This alkalinity enables rubidium carbonate to react with acids to form rubidium salts and carbon dioxide gas, a typical acid–base reaction of carbonates.
Rubidium carbonate is used mainly in specialized chemical research and niche technological applications. It serves as a precursor for the preparation of other rubidium compounds, including rubidium halides and rubidium hydroxide. In materials science and physics research, rubidium salts are used in studies involving optical materials, atomic spectroscopy, and quantum systems, where precise control of alkali metal species is required.
In certain cases, rubidium carbonate is used in glass manufacturing and specialty ceramics research, where alkali metal carbonates act as fluxing agents to modify melting behavior and material properties. However, sodium and potassium carbonates are far more commonly used for such industrial purposes due to cost and availability advantages.
From a physicochemical perspective, rubidium carbonate reflects typical trends in alkali metal chemistry. Compared with lighter alkali carbonates, it has a higher molar mass, greater ionic radius, and slightly different lattice energy characteristics. These factors influence its solubility and thermal behavior, although its overall chemical reactivity remains consistent with strong basic carbonates.
Overall, rubidium carbonate is a simple inorganic salt composed of rubidium and carbonate ions. While chemically similar to other alkali metal carbonates, its applications are largely confined to research and specialized fields involving rubidium chemistry, atomic physics, and materials science, where its unique isotopic and electronic properties are of interest.
References
2025. Thermal Structural Evolution and Optical Properties of Isosymmetric Nonlinear-Optical Borates α- and β-RbB3O5. Glass Physics and Chemistry.
DOI: 10.1134/s108765962560125x
2025. Potassium and Rubidium Thenoyltrifluoroacetonates: Crystal-Chemical Study and Thermal Properties. Journal of Structural Chemistry.
DOI: 10.1134/s0022476625100051
2022. COMPARATIVE CRYSTAL CHEMISTRY, SYMMETRY FEATURES, AND STRUCTURAL COMPLEXITY OF LiOH, NaOH, RbOH, CsOH, AND TlOH HYDROXIDES. Journal of Structural Chemistry.
DOI: 10.1134/s0022476622120174
|



















































 GHS07 Warning Details
GHS07 Warning Details