Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Anhui Lianchuang Biological Medicine Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lcywhx.com | |||
 | +86 (551) 6859-6228 6859-6338 +86 15856900656 | |||
 | +86 (551) 6859-6338 | |||
 | sales@lcywhx.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chongqing Xingcan Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.starrymed.com | |||
 | +86 (23) 6120-3303 +86 13650506873 | |||
 | +86 (23) 6120-3303 | |||
 | xingcanyaoye@sina.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Jinan Xinke Pharmaceutical Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xkpharm.net | |||
 | +86 13256107535 | |||
 | +86 (531) 8825-9693 | |||
 | haiyan.ma@xinkeyiyao.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zison Pharmaceutical (shandong)co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zisonpharm.com | |||
 | +86 (0531) 8825-9693 +86 15069083822 | |||
 | tracy.li@zisonpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Metabolism >> MAO inhibitor |
|---|---|
| Name | Safinamide mesylate |
| Synonyms | (S)-2-[[4-[(3-Fluorobenzyl)oxy]benzyl]amino]propanamide methanesulfonate |
| Molecular Structure | 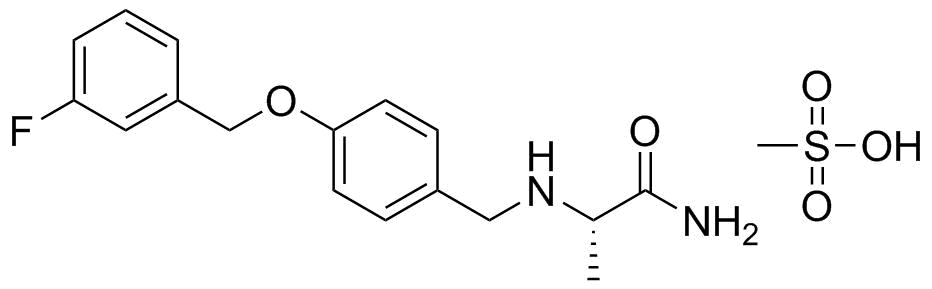 |
| Molecular Formula | C17H19FN2O2.CH4O3S |
| Molecular Weight | 398.45 |
| CAS Registry Number | 202825-46-5 |
| EC Number | 606-485-0 |
| SMILES | C[C@@H](C(=O)N)NCC1=CC=C(C=C1)OCC2=CC(=CC=C2)F.CS(=O)(=O)O |
| Hazard Symbols |    GHS05;GHS06;GHS09 Danger Details GHS05;GHS06;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H318-H400 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P273-P280-P301+P316-P305+P354+P338-P317-P321-P330-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Safinamide mesylate is the mesylate salt form of safinamide, a small-molecule drug used in the treatment of Parkinson’s disease. It functions as an antiparkinsonian agent with a dual mechanism of action, acting both as a reversible monoamine oxidase B (MAO-B) inhibitor and as a modulator of abnormal glutamate release. The mesylate (methanesulfonate) salt improves the compound’s stability and pharmaceutical formulation properties for oral administration. Safinamide was developed through medicinal chemistry programs aimed at improving symptomatic control of Parkinson’s disease beyond dopamine replacement therapy. Parkinson’s disease is characterized by progressive degeneration of dopaminergic neurons in the substantia nigra, leading to reduced dopamine levels in the striatum. Traditional treatments such as levodopa restore dopamine levels, but long-term use is associated with motor fluctuations and dyskinesias. Safinamide was designed to provide adjunctive therapy by enhancing dopaminergic signaling while also targeting non-dopaminergic mechanisms. The primary pharmacological action of safinamide is selective, reversible inhibition of monoamine oxidase B. MAO-B is an enzyme responsible for the breakdown of dopamine in the brain. By inhibiting MAO-B, safinamide increases extracellular dopamine levels in the striatum, thereby improving motor symptoms in Parkinson’s disease. Unlike irreversible MAO inhibitors, safinamide binds reversibly, which allows for more controlled pharmacological effects. In addition to MAO-B inhibition, safinamide has been shown to modulate voltage-gated sodium channels and reduce excessive glutamate release. This secondary mechanism is believed to contribute to its therapeutic effects on motor fluctuations and possibly on non-motor symptoms. Excessive glutamatergic activity has been implicated in excitotoxicity and motor complications in Parkinson’s disease, and modulation of this pathway provides an additional therapeutic benefit. The discovery of safinamide involved screening and optimization of compounds that could affect both dopaminergic and glutamatergic neurotransmission. The goal was to develop a multifunctional drug capable of addressing multiple pathological mechanisms in Parkinson’s disease. Structural modifications led to a molecule with appropriate central nervous system penetration, enzymatic selectivity, and pharmacokinetic properties suitable for once-daily oral dosing. Structurally, safinamide contains an aromatic core linked to an amide side chain and a fluorinated substituent that contributes to its binding affinity and metabolic stability. The mesylate salt form enhances its crystalline stability and solubility, which are important for consistent absorption and formulation in tablet form. Clinically, safinamide mesylate is used as an add-on therapy to levodopa in patients with Parkinson’s disease experiencing “off” episodes, which are periods of reduced motor control due to fluctuations in levodopa effectiveness. By increasing dopaminergic tone and modulating glutamate activity, safinamide helps extend “on” periods when motor symptoms are better controlled. Pharmacokinetically, safinamide is well absorbed after oral administration and undergoes hepatic metabolism, primarily through non-CYP enzymatic pathways, which reduces the potential for drug–drug interactions compared with some other antiparkinsonian agents. Its metabolites are eliminated mainly via renal excretion. Adverse effects associated with safinamide include dyskinesia, insomnia, nausea, and headache. These effects are generally related to increased dopaminergic activity and are typically manageable through dose adjustment or concomitant therapy modification. Overall, safinamide mesylate is a multifunctional antiparkinsonian drug that combines reversible MAO-B inhibition with modulation of glutamatergic transmission. Its development reflects a modern approach to Parkinson’s disease therapy, targeting both dopaminergic deficiency and downstream neurochemical dysregulation to improve motor symptom control in patients receiving levodopa therapy. References 2026. Development and Evaluation of Chitosan-Safinamide Mesylate Nanoparticles for the Management of Parkinson’s Disease. BioNanoScience. DOI: 10.1007/s12668-025-02347-x 2024. A novel stability-indicating chromatographic quantification of the antiparkinsonian drug safinamide in its pharmaceutical formulation employing HPTLC densitometry and ion-pair HPLC–DAD. BMC Chemistry. DOI: 10.1186/s13065-024-01315-y 2020. A combined theoretical and experimental study on the structure, vibrational, and electronic properties of antiparkinsonian drug safinamide. SN Applied Sciences. DOI: 10.1007/s42452-020-03661-7 |
| Market Analysis Reports |