Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| ZHIYU Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhiyubiotech.com | |||
 | +86 (512) 6279-1916 | |||
 | +86 (512) 6279-1915 | |||
 | sales@zhiyubiotech.com info@zhiyubiotech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Taizhou Tongxin Biopharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyrise.com | |||
 | +86 18652728585 | |||
 | +86 (523) 8276-5215 | |||
 | sales@allyrise.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ningbo Fengrui Fine Chemical Ltd. | China | |||
|---|---|---|---|---|
 | www.fengruichem.com | |||
 | +86 (574) 8772-0208 | |||
 | fr006@fengruichem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Alsachim SAS | France | |||
|---|---|---|---|---|
 | www.alsachim.com | |||
 | +33 (0) 390 402 200 | |||
 | +33 (0) 390 402 199 | |||
 | contact@alsachim.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Cell cycle >> CDK inhibitor |
|---|---|
| Name | Ribociclib |
| Synonyms | 7-Cyclopentyl-N,N-dimethyl-2-[[5-(1-piperazinyl)-2-pyridinyl]amino]-7H-pyrrolo[2,3-d]pyrimidine-6-carboxamide |
| Molecular Structure | 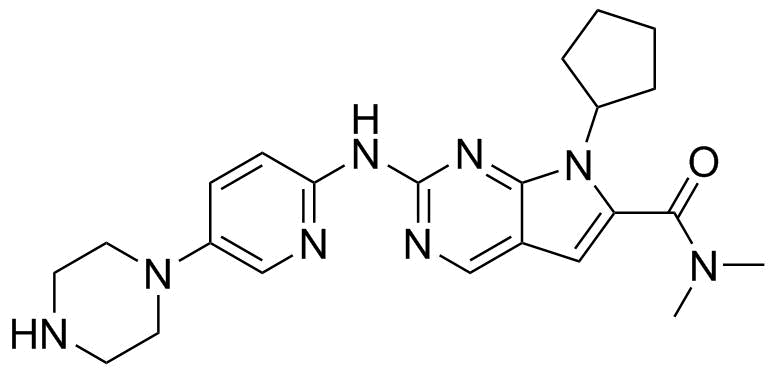 |
| Molecular Formula | C23H30N8O |
| Molecular Weight | 434.54 |
| CAS Registry Number | 1211441-98-3 |
| EC Number | 807-111-0 |
| SMILES | CN(C)C(=O)C1=CC2=CN=C(N=C2N1C3CCCC3)NC4=NC=C(C=C4)N5CCNCC5 |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 730.8$+/-$70.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 395.8$+/-$35.7 $degree$C (Calc.)* |
| Solubility | DMSO 90 mg/mL, Water $lessThan$1 mg/mL (Expl.) |
| Index of refraction | 1.723 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS08;GHS09 Danger Details GHS08;GHS09 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H360-H360FD-H361d-H373-H411 Details | ||||||||||||||||||||
| Safety Statements | P203-P260-P273-P280-P318-P319-P391-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Ribociclib is a small-molecule targeted anticancer drug that belongs to the class of cyclin-dependent kinase (CDK) inhibitors. It is specifically designed to inhibit CDK4 and CDK6, two enzymes that play a central role in regulating the cell cycle. Ribociclib is used in the treatment of certain hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancers. The development of ribociclib emerged from research into cell cycle regulation and the role of cyclin-dependent kinases in cancer proliferation. In normal cells, progression from the G1 phase to the S phase of the cell cycle is tightly controlled by CDK4 and CDK6 in complex with cyclin D. In many cancers, including hormone receptor-positive breast cancer, this regulatory pathway becomes overactive, leading to uncontrolled cell division. Ribociclib was developed to selectively inhibit CDK4/6 activity and restore control over cell cycle progression. Structurally, ribociclib is a heterocyclic small molecule containing a pyrrolopyrimidine core linked to substituted piperazine and cyclohexylamine moieties. These structural features enable the compound to bind competitively to the ATP-binding pocket of CDK4 and CDK6. By occupying this site, ribociclib prevents phosphorylation of the retinoblastoma protein (Rb), a key step required for progression from the G1 phase to the S phase of the cell cycle. When Rb remains unphosphorylated, it suppresses E2F transcription factors, thereby halting DNA synthesis and cell proliferation. The discovery of ribociclib was based on structure-guided drug design and optimization of kinase inhibitor scaffolds. Early screening efforts identified compounds capable of inhibiting CDK activity, and subsequent chemical modifications improved selectivity for CDK4/6 over other kinases. This selectivity is important for reducing off-target effects and improving clinical tolerability. Ribociclib was developed by Novartis as part of a broader effort to create targeted therapies for hormone-driven cancers. Ribociclib is administered orally and is typically used in combination with endocrine therapies such as aromatase inhibitors or selective estrogen receptor degraders. In hormone receptor-positive breast cancer, estrogen signaling drives cyclin D expression, which in turn activates CDK4/6. By inhibiting CDK4/6, ribociclib enhances the effectiveness of endocrine therapy and delays tumor progression. Clinically, ribociclib has been shown to improve progression-free survival in patients with advanced or metastatic breast cancer. It is approved for use in postmenopausal and premenopausal women, as well as in men, depending on regulatory guidelines. Treatment is generally administered in cycles to manage toxicity and maintain therapeutic efficacy. The pharmacokinetics of ribociclib involve oral absorption followed by hepatic metabolism, primarily mediated by cytochrome P450 enzymes, particularly CYP3A4. Drug–drug interactions are clinically relevant due to this metabolic pathway, and dosing adjustments may be required when used with other medications that affect CYP3A4 activity. Adverse effects associated with ribociclib include neutropenia, hepatotoxicity, and prolongation of the QT interval on electrocardiography. These effects are consistent with its mechanism of action on proliferating cells and its off-target influence on cardiac ion channels. Regular monitoring of blood counts, liver function, and cardiac rhythm is part of standard clinical management. Overall, ribociclib is a targeted CDK4/6 inhibitor developed to interfere with dysregulated cell cycle progression in hormone receptor-positive breast cancer. Its discovery reflects advances in understanding cancer biology and cell cycle control, and its clinical use represents a significant development in precision oncology, where therapy is guided by specific molecular mechanisms driving tumor growth. References 2026. The Impact of Conventional Chemotherapy Regimens and Targeted Drugs on Ovarian Function in Breast Cancer Patients. Reproductive sciences (Thousand Oaks, Calif.). DOI: 10.1007/s43032-026-02067-x 2026. Pediatric H3 G34-mutant diffuse hemispheric glioma: clinical, imaging and molecular prognostic factors, MGMT expression, and temozolomide response. Acta Neuropathologica. DOI: 10.1007/s00401-026-02992-w 2026. Real-world second- and third-line progression-free survival after progression on first-line CDK4/6 inhibitors in HR+/HER2- metastatic breast cancer by PAM50 intrinsic subtype: the SOLTI-1801 CDK-PREDICT study. Breast Cancer Research and Treatment. DOI: 10.1007/s10549-026-07931-7 |
| Market Analysis Reports |